Catia Contado* and Antonella Pagnoni
Department of Chemistry, University of Ferrara, Via Luigi Borsari 46, I-44123 Ferrara, Italy, E-mail: [email protected]
Introduction
Many ordinary cosmetics are now based on nano-emulsions and/or mineral-based nanomaterials. Colour, transparency, solubility and chemical reactivity are just a few of the properties altered with size and this is why so many manufacturers of cosmetics and personal care products are investing in research to create nanomaterials that are able to improve the quality of their products. Since their first use, benefits of nanotechnology have been widely publicised, but the discussion of the potential effects on their widespread use in consumer and industrial products is just emerging. The toxicity of the “traditional” chemical ingredients has been studied; a list of banned substances is included in the updated European Commission directive 76/768/EEC.1 However, there are still large gaps in our knowledge on the long-term effects in humans of chronic exposure to nanoparticles.
TiO2 has recently captured the attention of the scientific community since its safety assessment has been placed again under consideration. TiO2 is widely used as a sunscreen UV filter and as a colouring agent in all types of cosmetic products.2,3 TiO2 is employed as whitening and an anti-bacterial in toothpaste, as a component in stay-on cosmetics, to make pearl pigments for high quality eye and face make-ups, lipsticks, nail lacquers, and it is an important component also in many pharmaceutical products, as, for example, a colouring agent for capsules, films, tablets and coated pills, and medical sticky tapes. Initially, the Scientific Committee for Consumer Products (SCCP) declared TiO2 safe, and then in a more recent document4 it suggested a re-evaluation of TiO2, in the light of new scientific data on nano-sized particles.
Regulatory authorities have taken different positions regarding safety assessments of cosmetics; for example, the FDA (US Food and Drug Administration) assesses the safety of final products, while in the European Union (EU), safety assessment is mainly ingredient-based. From a regulatory perspective, ideally a nanomaterial in cosmetics should be characterised as it appears in the final formulation used by the consumer. Unfortunately, because of the limitations of existing analytical methodologies to achieve this, it seems, that the current best possibility for the safety assessment is to characterise an ingredient itself and then to take into account worst case considerations about its possible behaviour in the formulation. With this uncertainty, the need of analytical methods able to detect and monitor the presence of micro- or nano-sized particles inside of commercial products is a mandatory goal, not only to assess the quality of the ingredients but also to establish the safety of the finished products, particularly for the stay-on cosmetic products, which are subjected to a close, direct and prolonged contact with the skin and/or permeable mucous membranes (level of exposure).
Inductively-coupled plasma atomic emission spectrometry (ICP-AES), square wave voltammetry (SWV) and sedimentation field-flow fractionation (SdFFF) are described in this article to characterise and quantify the TiO2 particles inside six commercial foundation creams, purchased in Italy and the USA. The amount of TiO2 was determined on mineralised samples through SWV and ICP-AES, while the particle size distributions (PSDs) was determined on slurries with the SdFFF technique,5 which is a very powerful, non-destructive separation technique, suitable for separating colloids.6
Comparison of the ICP-AES and the SWV determinations: accuracy and precision
ICP-AES is a well-established technique which is known to give very rapid multi-elemental data. To quantify the TiO2 content in these types of commercial samples, an alternative technique is SWV at the hanging mercury drop electrode (HMDE). The new method set-up in this work was tested and critically compared with ICP-AES by analysing the TiO2 aeroxide powder (Degussa P-25), taken as the standard sample.
Six different amounts of TiO2 were sampled, accurately weighed and mineralised into clean, dry TFM (tetrafluoromethane) vessels, to which aliquots of concentrated HNO3, HF and H2O were added in sequence. The TFM vessels were sealed, placed in a high-pressure rotor and then in a microwave oven for which an optimised digestion programme was employed. At the end of the digestion programme, the samples were left to cool, transferred to a volumetric flask without filtering and diluted with deionised water. Each mineralised sample, diluted by a factor of 100, was analysed in triplicate by both ICP-AES and SWV for a total of 18 determinations for each method. The average results were almost identical [ICP-AES (av ± s) = (97.91 ± 2.19) TiO2 %w/w cf. SWV (av ± s) = (97.93 ± 2.79) TiO2 %w/w] and the “t” test assessed that the two methods were statistically equivalent at the 95% confidence level, even if the precision of the SWV determinations were lower than the corresponding value computed for ICP-AES. The ICP-AES and SWV average results were also compared with the 99.5 TiO2 %w/w declared by the manufacturer and the “t” test assessed that the two results were in agreement and statistically equivalent with the given purity (95% confidence level).
The SWV method also gave reliable and accurate results on a complex matrix such as that of a foundation cream. Two samples, labelled as #1 and #2, were considered for the test. Six samples of foundation cream were accurately weighed (about 0.1 g each) and mineralised by adding concentrated HNO3 and HF. At the end of the microwave digestion programme, the samples were left to cool and then transferred to a volumetric flask without filtering. Each mineralised portion was analysed in triplicate. SWV determinations were made by diluting the mineralised solutions with the supporting electrolyte solution (H2C2O4 0.25 M + H3BO3 0.20 M) by a factor of 100. The two methods gave statistically equivalent results for the mineralised foundation and also for these samples, ICP-AES gave more precise values: Sample #1 ICP-AES (av ± s) = (10.85 ± 0.24) %w/w cf. SWV (av ± s) = (9.67 ± 1.33) %w/w and Sample #2 ICP-AES (av ± s) = (8.03 ± 0.10) %w/w cf. SWV (av ± s) = (8.10 ± 0.44) %w/w.
TiO2 content in the six foundations: SWV and ICP-AES measurements
The six foundations chosen for this study were light beige in order to reduce the possible interference of other inorganic pigments, such as iron oxide in all its forms (CI 77491 red, CI 77492 yellow, CI 77499 black). Sample #1 was the only sample among those considered in this study in which TiO2 was listed among the ingredients as well as “possibly present”; samples #1 and #4 had a sun protection factor (SPF) of 15 and 18, respectively.
SWV determinations were made in triplicate, by diluting the mineralised solutions with the supporting electrolyte solution (H2C2O4 0.25 M + H3BO3 0.20 M) by a factor of 10 or 100, depending on the sample. The highest content of TiO2 (~10.82 %w/w) was confirmed for sample #1, while sample #6 had the lowest (~5.24 %w/w), closely followed by sample #4 (~5.48 %w/w); see Table 1.
Table 1. %TiO2 content: SWV and ICP-AES determinations only on the mineralised samples. ICP-AES reading λ = 336 nm for Sample #2, 334 nm for all the others.
Sample | #1 | #2 | #3 | #4a,b | #5 | #6a,b |
Weighed amount of foundation (g) | 0.1119 | 0.1085 | 0.1114 | 0.1778 | 0.1187 | 0.1549 |
0.1039 | 0.1336 | 0.1295 | 0.1162 | 0.1028 | 0.1439 | |
0.1303 | 0.1339 | 0.1145 | 0.1606 | 0.1730 | 0.1535 | |
SWV: | ||||||
TiO2 Average (w/w%) | 10.82 | 8.10 | 9.15 | 5.48a | 6.73 | 5.24 |
SdFFF (w/w%) | 0.30 | 0.44 | 0.44 | 0.63 | 0.26 | 0.58 |
RSD % | 2.8 | 5.5 | 4.8 | 11 | 3.9 | 11 |
ICP-AES: | ||||||
TiO2 Average (w/w%) | 11.03 | 8.03 | 9.92 | 8.007 | 7.35 | 7.37 |
SdFFF (w/w%) | 0.21 | 0.10 | 0.23 | 0.041 | 0.28 | 0.70 |
RSD % | 1.9 | 1.3 | 2.4 | 0.51 | 3.8 | 9.6 |
aProbably presence of interfering elements, such as Pb2+ (ultra-trace) on the right and Cu2+ (trace) on the left of the SWV Ti4+ peak.
bMineralised solution clear but yellow
RSD, relative standard deviation.
The TiO2 content, determined through ICP-AES, ranged between 11.03 %w/w (sample #1) to 7.35 %w/w (sample #5); see Table 1. The ICP-AES values were usually higher than the corresponding SWV data; the only exception was sample #2, for which the two techniques gave very similar results; ICP-AES also gave more precise determinations in this series of experiments.
The two methods, compared for each sample at the 95% confidence level, were statistically equivalent for samples #1–3, while for those samples in which interfering substances were found, only the ICP-AES results were considered reliable.
Foundation cream: TiO2 particle solvent extraction for the size analyses
A representative portion of TiO2 particles was isolated from the six foundations by applying a solvent extraction procedure with a ternary mixture of solvents: (water/methanol/hexane in the volume ratio 1 : 4 : 1).7 The most suitable ratio among the solvents was determined through a set of preliminary experiments in which different volumes of water, methanol and hexane were mixed. The procedure can be summarised as follows: a weighed amount of foundation was dispersed in water and mixed for 60 s with an ultrasound probe set at 50% of maximum nominal power; after the addition of methanol, the suspension was mixed again for 60 s with the ultrasonic probe and transferred into a separation funnel to which the hexane was added; the suspensions were vigorously shaken to ensure good mixing and left to equilibrate for 1 h; then the two phases were separated. The aqueous phases (slurries) were kept for mineralisation, the ICP-AES determinations, the SdFFF separations and transmission electron microscopy (TEM) observations. Since the methanol might switch off the torque during the ICP-AES analysis performed on the slurries or it could be an interfering agent during the mineralisation process, it was always removed from the samples by evaporation under N2 atmosphere and the lost volume substituted with deionised water. When concentrated sample slurries were necessary, the methanol was quickly eliminated by evaporation at 70°C.
SdFFF–ICP-AES analysis
The six concentrated suspensions were fractionated with the SdFFF technique under field programmed conditions. The particle size distribution (PSD), computable by elaborating the fractograms, required the knowledge of the sample density, which was inserted as an “input parameter”. In this case, it was set to 4.05 g mL–1, the average value of the two most common crystallographic phases of TiO2, rutile and anatase, the densities of which are 3.84 g mL–1 and 4.26 g mL–1, respectively.8 All fractograms presented very intense void time peaks due to particles of mass which cannot be fractionated by SdFFF, i.e. particles with sizes lower than 100 nm or higher if their density is lower than ~4 g mL–1.
During the separation process, fractions of 6 mL each were collected for the off-line ICP-AES determinations. The Ti detection limits were in the range 17–95 µg L–1, depending on the sample, so that the concentration determined in the fractions was sometimes very close to these values; however, even if the data could not be considered quantitative, the important aspect was the correlation between the UV elution profile and the elemental composition of the eluate.
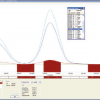
Examples of the separation results are shown in Figures 1 and 2, in which the Ti concentration values, expressed in µg L–1, are reported on the right secondary y-axis of graph (a). Figure 1, the analysis of sample #3, presents an UV elution profile with two partially overlapped peaks, the first at about 30 min and the second at about 40 min, which the correspondent PSD assigns to particles of about 230 nm for the first and of 300 nm for the second. The Ti concentration profile peak perfectly matches only the second population of particles. The TEM pictures of this sample, reported in the figure, show two different types of particles, both of regular shape (roundish), observed also in samples #1 and #5; those of higher dimensions have a lighter, almost transparent texture to TEM, typical of a triple emulsion state, for which the assumption of a density of 4.05 g mL–1 is surely inappropriate.


The suspension of sample #6, the analysis of which is reported in Figure 2, was opalescent white and very stable in time, requiring a stronger initial SdFFF field to be fractionated. The fractogram has a very narrow peak at about 8 min, immediately after the very intense void peak and then a profile that slowly decreases almost to the base line within 60 min; in the PSD plot (b), the first peak falls at about 90 nm, then the particle population spans up to 400 nm. The ICP-AES determinations give a Ti profile which perfectly matches the first narrow peak, but does not follow the UV trace towards the end of the fractogram, where the Ti concentration increases but only a few particles (~260 nm) elute. The TEM photographs reported in (c) and (d) confirm that this sample contains mainly very small and compact particles of irregular shape ranging between 60 nm and 130 nm and a consistent amount of small needle particles, which are peculiar to this sample.
Conclusions
The TiO2 contained in commercial foundations can be determined through the consolidated ICP-AES spectroscopic technique but to contain the costs, the SWV can be used as an alternative method since the results on the mineralised samples agreed and were achieved with a good precision and accuracy.
A higher amount of TiO2 was found in sample #1, commercialised in the USA, for which the TiO2 was declared as an ingredient (~11 %w/w), while all samples commercialised in Europe (Italy) had a lower TiO2 content (in most cases <8 %w/w).
The SdFFF technique was used to size the particles contained in the commercial foundations. The Ti off-line ICP-AES slurry determinations, made on collected fractions during the fractionation, allowed TiO2 to be classified as pigment, i.e. with particles of sizes higher than 100 nm. However, in all considered samples, a large number of nanoparticles were detected, the chemical nature of which should be further investigated.
References
- European Commission, Consumer Affairs: Consolidated version of Cosmetic Directive 76/768/EEC. http://ec.europa.eu/enterprise/sectors/cosmetics/documents/directive/ (Accessed July 2011).
- European Commission, Scientific Committee on Consumer Products: SCCP/1147/07 Opinion on Safety of Nanomaterials in Cosmetic Products. http://ec.europa.eu/health/ph_risk/committees/04_sccp/docs/sccp_o_123.pdf (Accessed July 2011).
- http://eur-lex.europa.eu/LexUriServ/site/en/consleg/1976/L/01976L0768-20061003-en.pdf (Accessed July 2011)
- Guidance Note: The “Restricted Fields of Application” for Coloring Agents in Annex IV to Cosmetics, Directive 76.768/EEC (Accessed July 2011). http://ec.europa.eu/enterprise/sectors/cosmetics/files/doc/colouring_agents_restricted_fields_of_appl_en.pdf
- M.E. Schimpf, K.D. Caldwell and J.C. Giddings (Eds) Field-Flow Fractionation Handbook. Wiley-Interscience, New York, USA (2000).
- C. Contado and A. Pagnoni, Anal. Methods 2, 1112–1124 (2010).https://doi.org/10.1039/c0ay00205d
- C. Contado and A. Pagnoni, Anal. Chem. 80(19), 7594–7608 (2008).https://doi.org/10.1021/ac8012626
- M.J. O’Neil (Ed) The Merck index: An Encyclopedia of Chemicals, Drugs, and Biologicals, 14th Edn. Merck Research Laboratories: Whitehouse Station, NJ, USA (2006).