Paul D.A. Pudney
Unilever Discover, Colworth, Colworth Science Park, Sharnbrook, Bedfordshire, MK44 1LQ, UK
Skin is the largest organ of the human body and accounts for 16% of our body weight with approximately 1.6 m2 of surface area. Without skin, terrestrial life would be impossible. Skin acts as a semi-permeable membrane that helps prevent too much water loss that would otherwise lead to desiccation and death. It also stops unwanted external threats such as chemical insult, invasion of microorganisms, mechanical injury and ultraviolet light harming the body. This is the so-called “outside in-inside out” paradigm for the function of skin. The main barrier part of the skin has long been identified as being the stratum corneum (SC), which is the outer 20 µm or so of the skin. It is a heterogeneous structure which is usually described as being bricks and mortar, with flattened coenocyte cells as the bricks surrounded by lipid mortar. It is a highly effective barrier. Originally thought as a dead layer, it has gradually been found to have substantial metabolic activity and acts like a smart responsive material. It is a dynamic, structurally intricate membrane.
Thus, understanding the SC’s function and structure has implications in many areas of health and industry. If it malfunctions it can cause many diseases, some of which are very prevalent. For example, there are eight million people living with a skin disease in the UK alone. Some are manageable, others are severe enough to kill. These include different types of cancer to others such as Eczema (Atopic Eczema), Psoriasis and acne. Also the pharmaceutical industry, for instance, is interested in delivering drugs across the skin to avoid the digestive system. The cosmetic industry are interesting in improving the SC’s health and appearance by moisturising and targeting skin actives and measuring their effect. Thus, there is great need for methods to be able to characterise the structure and its changes.
There are many spectroscopic methods that can contribute to understanding, but one that has been claimed to be at the forefront of skin research is Raman spectroscopy. This article will concentrate on its use. Considering that the first full Raman spectrum of skin was not published until 1992 by Edwards et al.,1 its progress has been remarkable. It was realised, as with other biological systems, that Raman spectroscopy has many inherent advantages. Its molecular sensitivity tells us not only which molecules are present but, in many cases, is able to say much about their state or order. Also, its relative lack of sensitivity to water allows the other molecules to be seen in the spectrum (c.f. infrared spectroscopy); however, it can also be used to measure hydration levels of the skin. Further, because it is a scattering-based method the confocal principle can be used, hence allowing non-invasive depth profiling. Thus, not only does it have all the requirements to look at skin, but also potentially non-invasively and in vivo.
The biggest impact of Raman spectroscopy in skin research has been with in vivo studies, with the main set-up usually following that first developed by Caspers and Puppels.2 This is essentially an inverted microscope with an optical flat window for the skin to be placed on, Figure 1.

This maintains a clear optical path with refractive index matching, thus giving good depth resolution and high signal levels. Having very good signal-to-noise in in vivo experiments is even more important than normal, as you are asking subjects to be measured for sometimes significant periods of time and minimising this is crucial. There are variants of this set-up, but here we will concentrate on the only commercially available system available (from RiverD international) with which most studies are now done. It comprises a high-performance dispersive spectrometer with 671 nm and 785 nm laser excitation and a confocal measurement stage. The spectrometer allows for measurements in the so-called high-wavenumber region (2400–4000 cm–1) and the fingerprint region (400–2400 cm–1) of the Raman spectrum.
To further describe the experiment, normally the laser would be focussed just above the skin surface in the optical flat and then incremental steps would be taken down through the SC into the viable epidermis, normally around 30 µm in total, but it is possible to go deeper. This allows the position of the surface of the skin to be calculated accurately and then to pass through the SC into the viable epidermis; the depth of which can also be calculated from the data. This can be applied to many problems and this article will show three of the main ones: measuring the inherent components of the SC and how they can change, measuring the hydration levels and, finally, how penetration of active molecules can be followed.
Composition of the skin and its changes
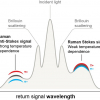
The initial work on this was done by Gerwin Puppels et al. in the late 1990s. A Raman spectrum from the SC is complicated and arises from the many individual components within it. These components are, however, well known and mainly comprise of keratin (the main structural protein), water, lipids (of which ceramides are the main class), natural moisturising factors (NMF) and molecules from sweat. NMFs are the breakdown product of the important skin protein, filaggrin (FLG), and consist mainly of a range of hydroscopic amino acids. They are important for regulating water content, pH and elasticity of the SC. The analysis approach taken is to model the spectra obtained from the SC with the spectra of the main individual components present: this method works well in most circumstances. Thus, when Raman spectra are collected as a depth profile confocally through the SC, each spectrum can be modelled to give a depth profile of the main components of the SC. The usual ones obtained from the fingerprint region are keratin, NMF, ceramide 3, cholesterol, urea, lactic acid (pH4) and trans-urocanic acid. Further, using the high wavenumber region, a profile of the water content can be obtained. This is determined from the ratio of Raman intensities of the OH stretch vibration of water at 3390 cm–1 and the CH3 stretch of protein at 2935 cm–1. Raman spectroscopy thus can measure all the crucial elements that make up the SC with depth, in vivo. Given the demonstration that this can be done, multiple applications can be envisaged. Here we illustrate some of the main types of study that have been completed to clearly show the power of the method.
Atopic dermatitis (AD)
Atopic dermatitis is an increasingly common type of dermatitis also known as atopic eczema; it is an inflammatory, relapsing, non-contagious and itchy skin disorder. It has been shown that single mutations in the gene for filaggrin are a predisposing factor for AD in 50% of cases. Filaggrin is a key protein in the formation of the SC and, as discussed above, it is the source of the hydroscopic amino acids known as NMFs that help control SC hydration. In a large study,3 149 subjects were tested for polymorphisms in the filaggrin gene, then measured using in vivo Raman spectroscopy to obtain their NMF levels. A summary of the results are shown in Figure 2.

As can be clearly seen, there is significant reduction of NMF levels in carriers of filaggrin loss-of-function mutations. This result is true for all depths within the SC. It was further shown within patients with AD, that Raman signatures of NMF level predict FLG mutation status, overcoming the need for the more technically demanding genotyping. It also helped to further inform the work to unravel the complex nature of interplay of different factors involved in AD.
Different skin types: axilla and scalp (with and without dandruff)
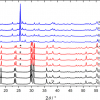
Although skin covers most of the body, it is known not to be uniform; this is most likely so it can cope with the different stresses on anatomically different areas. One of the observed variations is the number of cell layers and hence thickness of the SC. Palmo-plantar skin, for instance, is especially thick and has a number of well-known differences. Other body sites which show some evidence of differences include the axilla and the scalp skin. These areas are difficult to access using the standard configuration of the in vivo Raman instrument, hence a new smaller pen-shaped probe has been developed, see Figure 1. It allows the window to be placed against the subject in more curved and recessed areas of subject’s body and also for the subject to be more comfortable whilst the measurements take place. Using this probe the axilla and scalp can be studied. The scalp and axilla SC show significant differences from the “normal” SC of the volar forearm. For instance, the scalp is observed to have lower amounts of NMFs compared to the volar forearm within the same subjects. This is at a similar lower level as was observed with the subjects’ mutations in the gene for filaggrin described above, although clearly the cause is different. Further, for both the axilla and scalp the lipids show a change in order as compared to the lipids in the volar forearm and also differences from each other.4 These Raman spectroscopy results clearly add to the present knowledge of the structure and function of different skin areas of the body. Using the same Raman probe, a comprehensive study of dandruff and the effect of anti-dandruff shampoo with zinc pyrithione (ZnPTO) has been completed.5 This identified a number of changes in the dandruff scalp SC, including a further reduction of the NMF levels to 0.16 au compared with 0.39 au in healthy scalp: normal forearm levels are above one. Treatment with the anti-dandruff shampoo shows restoration of these levels to those observed in healthy scalp, see Figure 3. This was also true of other components in the scalp, which were shown to be lower in dandruff sufferers.

Skin hydration
SC water content is known to reflect skin health and is related to changes in elasticity, flexibility, surface morphology and sensory feeling. The water content across the SC obtained in depth profile by in vivo Raman spectroscopy is accepted as a standard, reliable method. This depth profile is also used to determine the SC thickness, as the water content is low on the outside and reaches a plateau as you move into the viable epidermis. What is more controversial, however, is how to get the SC thickness from the water profile. Many analysis methods have been suggested, but, as the profile shape changes with different levels of hydration and different skin thickness, the different models perform with great variability. A recent study by Tom Hancewicz et al.6 showed that a consensus approach to the problem—where a number of different methods were used and averaged—was found to be necessary in order to properly model and quantify the large diversity of water profile types encountered in typical in vivo Raman spectroscopy water measurement. These results were then validated against reflectance confocal microscopy measurements. This makes in vivo confocal Raman spectroscopy, with its unique advantage of non-invasive, spatially resolved depth profiling of skin hydration, the best method to study the range of medical, cosmetic and pharmaceutical problems where skin hydration is at their heart.
Penetration of actives into skin
The pharmaceutical and cosmetic industries are both interested in how molecules penetrate the skin. The pharmaceutical industry primarily to avoid any complications the gastrointestinal tract may have on a drug. For both industries, if the target is the skin itself, locating how and where an active molecule penetrates into skin is fundamental in understanding any benefit it may impart. Thus, this is a very good application for in vivo Raman spectroscopy. A comprehensive study on the skin active, retinol, illustrates well what can be done; it has been studied from a range of delivery vehicles and with and without penetration enhancers.7 It shows when and how much, quantitatively, retinol penetrates through the SC and into the viable epidermis, the target tissue. The profound effect of penetration enhancers, like oleic acid and triton 100, was also characterised. As Raman spectroscopy measures all components, not only the active molecule, it also shows how far the vehicle itself has penetrated and it was seen to be ahead of the active molecule in all cases.
A recent study has compared in vivo penetration with in vitro,8 the vast majority of studies of skin penetration are done in vitro and are assumed to be representative of the in vivo situation. There are very few reported correlations of such in vitro studies with in vivo data. Raman spectroscopy is the ideal method to do this. This study used the model active, niacinamide, in a range of vehicles. It found a good correlation between amount of niacinamide permeated through skin in vitro to the intensity of the niacinamide signal determined in the stratum corneum in vivo. Also, as in the retinol study, the way the vehicle penetrated was crucial to the amount of active that was delivered. It also highlighted the need for many more studies of this type to help further validate the in vitro work as well as understand the skin penetration process in more mechanistic detail. Thus, in vivo Raman spectroscopy is poised to make an even more significant contribution to this high important area of skin research.
Summary
The examples show a wide range of application for in vivo Raman spectroscopy have now been realised. It is technique that is expanding its range and being used ever more widely. It is clearly in the forefront of skin research today.
References
- B.W. Barry, H.G.M. Edwards and A.C. Williams, J. Raman Spectrosc. 23, 641 (1992). doi: http://dx.doi.org/10.1002/jrs.1250231113
- P.J. Caspers, G.W. Lucassen, R. Wolthuis, H.A. Bruining and G. J.Puppels, Biospectroscopy 4, S31 (1998). doi: http://dx.doi.org/10.1002/(SICI)1520-6343(1998)4:5+3.0.CO;2-M
- S. Kezic , P.M.J.H. Kemperman, E.S. Koster, C.M. de Jongh, H.B. Thio, L.E. Campbell, A.D. Irvine, I.W.H. McLean, G.J. Puppels and P.J. Caspers, J. Invest. Dermatol. 128, 2117–2119 (2008). doi: http://dx.doi.org/10.1038/jid.2008.29
- P.D.A. Pudney, E.Y.M. Bonnist, P.J. Caspers, J.-P. Gorce, C. Marriot, G.J. Puppels, S. Singleton and M.J.G. van der Wolf, Appl. Spectrosc. 66, 882–891 (2012). doi: http://dx.doi.org/10.1366/12-06640
- E.Y.M. Bonnist, P.D.A. Pudney, L.A. Weddell, J. Campbell, F.L. Baines, S.E. Paterson and J.R. Matheson, Int. J. Cosmetic Sci. 36, 347–354 (2014). doi: http://dx.doi.org/10.1111/ics.12132
- T.M. Hancewicz, C. Xiao, J. Weissman, V. Foy, S. Zhang and M. Misra, J. Cosmet. Dermatol. Sci. Appl. 2, 241–251 (2012).
- M. Mélot, P.D.A. Pudney, A.-M. Williamson, P.J. Caspers, A. Van Der Pol and G.J. Puppels, J. Control. Release 138, 32–39 (2009). doi: http://dx.doi.org/10.1016/j.jconrel.2009.04.023
- D. Mohammed, P.J. Matts, J. Hadgraft and M.E. Lane, Pharmaceut. Res. 31, 394–400 (2014). doi: http://dx.doi.org/10.1007/s11095-013-1169-2